Acidosis Has Two Stages in the Body
A Terrain-Based Understanding of Acid-Base Balance
Your body is naturally designed to regulate its internal pH with great precision. The blood must remain slightly alkaline — around 7.35–7.45 — to sustain life. But while the blood pH is tightly controlled, other parts of the body, particularly the fluid that surrounds your cells (interstitial fluid or “terrain”), are more vulnerable to subtle shifts in acid-base balance.
In this model, we explore two stages of acid stress in the body:
—
Stage 1: Latent (Tissue-Level) Acidosis
This first stage is subtle and not picked up by blood tests.
In this stage, waste products from metabolism, poor diet, and lifestyle begin to accumulate in the fluid between your cells. The lymphatic system, kidneys, and lungs work hard to remove these acids — but when they’re overburdened or lack the needed cofactors (like minerals and hydration), acids may temporarily accumulate in tissues.
This adaptive state may not disrupt blood pH, but it can silently affect:
Mitochondrial energy production
Inflammation and immune function
Enzyme activity and detox processes
Bone mineral balance
Cellular signaling
Over time, this may contribute to the terrain imbalances underlying chronic disease, including metabolic syndrome, joint degeneration, neurological disorders, and autoimmune issues.
—
Stage 2: Metabolic (Blood-Level) Acidosis
If the terrain remains overloaded for too long, the body can no longer keep the acid burden out of the bloodstream. This leads to metabolic acidosis, a more serious condition where blood pH drops below safe levels.
This stage is recognized by conventional medicine and can result from:
Kidney failure
Diabetes (ketoacidosis)
Shock or severe illness
Drug or toxin overload
This stage is dangerous and requires medical intervention. But from a New Biology view, it’s often preceded by years of adaptive buffering — where the body tries to protect the blood by storing acids in tissues.
—
⚖️ What Contributes to Acid Terrain Stress?
Latent acidosis isn’t caused by meat or protein alone — it’s the result of overload and deficiency combined:
Mineral-deficient diet
Processed, denatured foods
Overeating or food out of sync with season
Chronic stress, shallow breathing
Dehydration, poor water quality
Lack of movement, sun, and sleep
Toxic exposure or poor lymphatic flow
—
Buffering: The Body’s Natural Coping System
To keep blood pH stable, your body “borrows” buffers:
Calcium and phosphate from bone
CO₂ regulation through breath
Bicarbonate from the kidneys
Proteins and minerals from food and tissue
But these resources are finite. Without nutrient-dense, mineral-rich food and natural living habits, the buffering system becomes strained.
—
Terrain Healing = Mineral Replenishment + Drainage
Instead of counting “acid” and “alkaline” foods, focus on what the terrain needs to eliminate waste and restore energy:
✅ Spring or structured water
✅ Sea salt, kelp, dulse, leafy greens
✅ Raw dairy, bone broth, organ meats
✅ Movement, breathwork, grounding
✅ Time outdoors and exposure to natural light
✅ Rest, warmth, and emotional balance
—
Remember
Your blood may always appear “normal,” but that doesn’t mean your terrain is thriving.
Healing starts at the cellular level — in the fluid between your cells.
This is the true terrain where chronic disease begins and where it can be reversed.
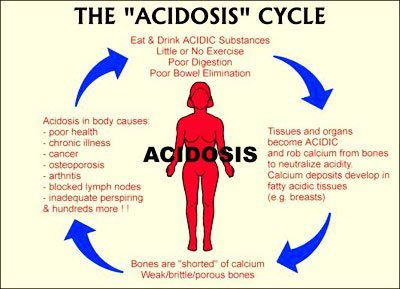

Neutralising a high acid load with proper dietary and lifestyle practices might just be the key to unlocking great health & preventing disease.
- Kidneys filter and secrete acids, needing alkaline foods to do this. The kidneys produce bicarbonate ions, which neutralize acid within the blood.
An analogy of this might be living in a cabin in the arctic, which has a wood fired stove. The stove can keep you warm as long as there is enough fire wood. When you run low on fire wood, you begin rationing it to. In other words, the kidneys and the lungs can only balance the pH of the blood and maintain an alkaline interstitial fluid (extracellular fluid) for as long as they have sufficient alkaline nutritional co-factors.
Discover more from Feel Good Counselling
Subscribe to get the latest posts sent to your email.
